In the ever-evolving landscape of women’s health, polycystic ovary syndrome stands as one of the most prevalent yet enigmatic conditions affecting reproductive-age individuals. As 2025 draws to a close, a wave of groundbreaking studies has illuminated new paths toward better diagnosis, management, and potentially even prevention. From genetic discoveries to subtype classifications, these developments promise to transform how healthcare providers approach this multifaceted disorder, offering hope to the estimated 1 in 10 women worldwide who grapple with its effects.
This year has seen researchers delve deeper into the biological underpinnings, revealing connections that extend beyond traditional symptoms. Emerging evidence suggests the syndrome may be part of a larger metabolic framework, influencing not just women but potentially men as well, through shared genetic and environmental factors. Such insights are reshaping clinical guidelines and opening doors to personalized medicine.
Understanding the Basics
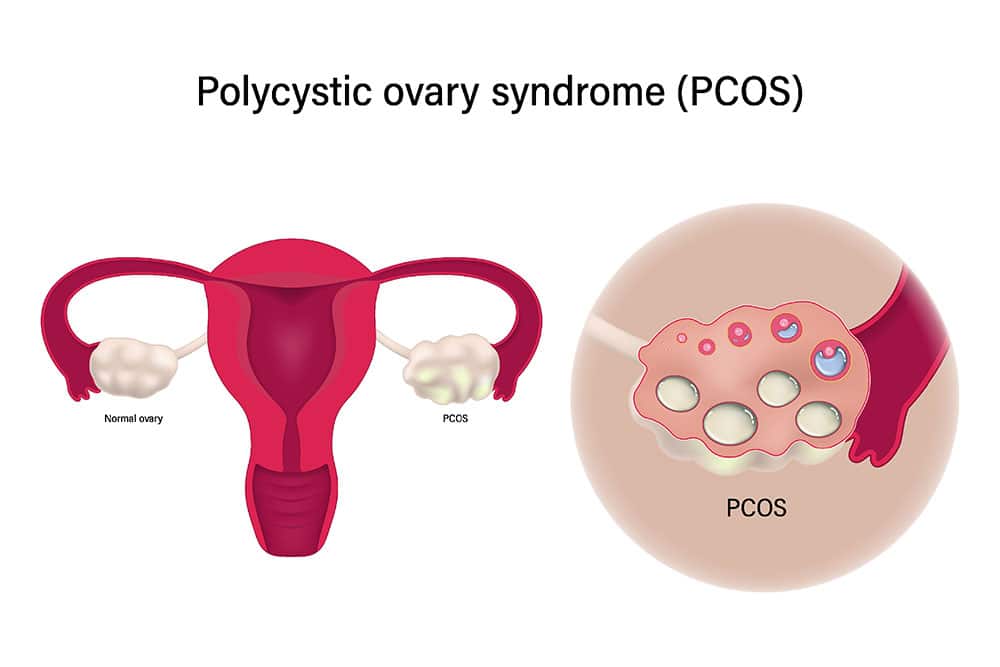
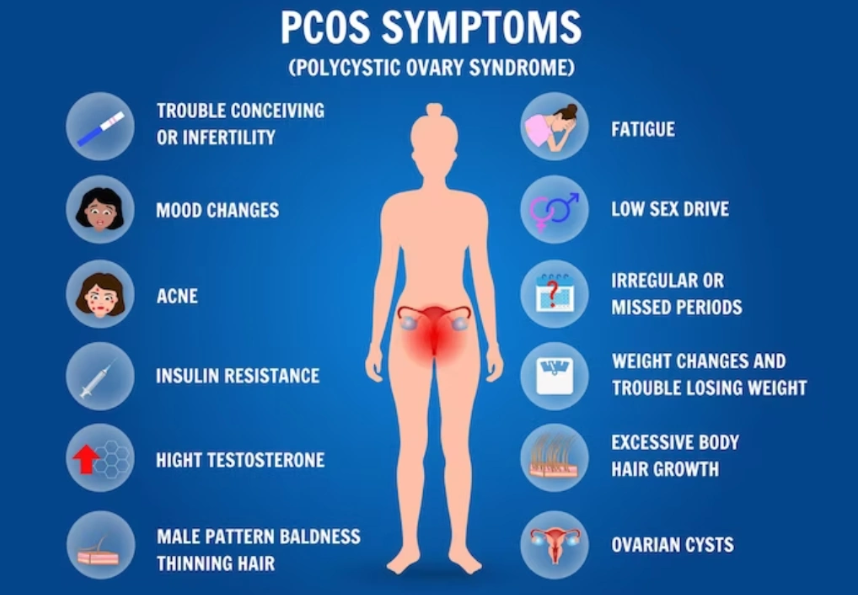
At its core, the condition involves a hormonal imbalance that can lead to irregular menstrual cycles, elevated levels of male hormones, and the formation of small cysts on the ovaries. Common manifestations include weight gain, acne, excessive hair growth, and fertility challenges. Beyond these, it heightens risks for long-term issues like type 2 diabetes, heart disease, and mental health concerns such as anxiety and depression.
According to health authorities, metabolism problems often accompany hormonal disruptions, impacting overall well-being and appearance. Early detection is crucial, particularly during adolescence, to mitigate these risks through lifestyle interventions and medical support.
While the exact cause remains elusive, a combination of genetic predisposition and environmental influences is believed to play a role. Family history often emerges as a key indicator, prompting experts to advocate for routine screening in at-risk populations.
Genetic Breakthroughs Offer Clues to Risk Factors
One of the most significant strides this year came from the largest genetic analysis conducted to date. Scientists examined the genomes of over 440,000 women across Chinese and European ancestries, including 25,000 diagnosed cases. This effort uncovered 94 genetic variants associated with increased susceptibility, 73 of which were previously unidentified.
These variants account for approximately 27% of risk variation in European groups and 34% in Chinese populations. Notable findings include alterations in genes related to mitochondrial function, sex hormone-binding globulin levels, and ovarian cell activity. For instance, a variant in the mitochondrial ribosomal protein S22 gene aligns with prior observations of mitochondrial dysfunction in affected individuals.
The implications are profound: with genetics contributing to about 70% of overall risk, these discoveries could pave the way for targeted therapies. Researchers highlighted potential drugs like clomifene, already used for fertility, and betaine, which might address specific pathways. This shift from symptom management to addressing root causes could revolutionize care, emphasizing the need for diverse genetic studies to ensure equitable advancements.
Identifying Subtypes for Tailored Approaches
Another pivotal development is the classification of the syndrome into four distinct subgroups based on clinical data. This data-driven approach uses commonly measured variables to differentiate phenotypes, enabling more precise interventions.
The subgroups include:
- Hyperandrogenic Phenotype: Marked by high male hormone levels, this group faces elevated miscarriage risks and lipid abnormalities but responds well to certain IVF techniques.
- Overweight-Obesity Phenotype: Characterized by higher body mass index and insulin resistance, it presents the most severe metabolic challenges but also the highest recovery potential.
- High Sex Hormone-Binding Globulin Phenotype: A milder variant with fewer fertility hurdles and lower risks for diabetes and hypertension.
- Elevated Luteinizing Hormone and AMH Phenotype: Associated with risks during fertility treatments but distinct in its hormonal profile.
These distinctions highlight variations in pregnancy outcomes and complications, underscoring the value of precision medicine. For example, subgroup-specific IVF protocols could improve success rates while minimizing adverse effects.
Linking DNA Variants to Hormone Production
A collaborative study between Mount Sinai and Duke University has pinpointed specific DNA variants in the DENND1A gene that drive excessive testosterone production, a hallmark feature. Using advanced techniques like high-throughput assays and CRISPR editing, researchers identified regulatory elements across 14 genomic regions linked to the condition.
In human cell models, activating these variants boosted DENND1A expression and testosterone levels, providing the first direct evidence of inherited factors altering hormone output. This breakthrough not only validates genetic influences but also suggests avenues for novel therapeutics that could modulate these pathways, potentially alleviating symptoms at their source.
Innovations in Diagnosis and Management
2025 has also brought updates to clinical guidelines, recognizing the syndrome as a risk factor for cardiovascular events even in younger years. Antimüllerian hormone (AMH) has gained prominence as a biomarker, replacing more invasive methods like antral follicle counts for diagnosis.
Furthermore, explorations into the gut microbiome reveal alterations that may contribute to inflammation and metabolic issues. This connection to mental health disorders opens innovative treatment strategies, such as microbiome-targeted therapies, to complement existing options like lifestyle changes and medications.
Ongoing trials, such as the REBALANCE Study focusing on infertility, are recruiting participants to test new interventions. These efforts emphasize patient-centered care, addressing not just physical but also psychological aspects.
Broader Implications and Future Directions
The evolving perspective views the syndrome within a wider metabolic context, potentially affecting male relatives through similar mechanisms. This holistic approach encourages family-wide screenings and preventive measures.
Experts stress the importance of awareness, as many cases remain undiagnosed. Physical changes can impact self-esteem, though the condition itself doesn’t directly cause mental disturbances—rather, it’s the societal and personal responses to these changes that matter.
As research progresses, the focus shifts toward comprehensive strategies that integrate genetics, subtypes, and innovative diagnostics. This could lead to earlier interventions, reducing long-term health burdens and improving quality of life.
Resources for Support
For those seeking more information, the Office on Women’s Health provides detailed overviews and guidance. Additionally, explore specialized insights on PCOS through Aspect Health’s dedicated platform.
In conclusion, 2025 marks a turning point in our comprehension of this complex condition. With continued investment in diverse research and collaborative efforts, the future holds promise for more effective, individualized solutions. As awareness grows, so does the potential to empower those affected to lead healthier, fuller lives.
